Research
In the etzoldlab
The challenges arising with the needed global energy change as also future sustainable feedstock supply for chemical industry is major guideline for the research in the etzoldlab. From the perspective of chemical engineering the research carried out bridges from the molecular scale and catalysis development, over the scale of transport pathways, e.g. within gas-diffusion electrodes, towards the reactor/electrolyser scale. Finally, the influence of changes on these different scales on full chemical processes is assessed through simulation approaches. Thus, catalyst synthesis and electrode design go hand in hand with experimental testing at technical relevant conditions as well as chemical reaction engineering and flow sheeting simulation. As a future sustainable energy and chemical industry will need a concerted interaction of electrochemical and classical heterogeneous catalysed processes both are studied. Based on this strategy the research of the etzoldlab can be divided in three strongly interacting sub-groups. More details on the research of these subgroups can be found in the sections below.
With our research we are part of the following huger or collaborative research activities:
- Research Field E+E: Energy and Environment
- Profile Topic Carbon-Neutral Circles
- Clean Circles – Iron as energy carrier in a carbon neutral circular energy economy
- CRC 1487 Iron, upgraded!
- LOEWE research cluster FLOW FOR LIFE
 ERC Consolidator Grant IL-E-CAT
ERC Consolidator Grant IL-E-CAT
- STA 449F3 Jupiter coupled with mass spectroscopy and automatic sample changer for oxygen-sensitive measurements (Netzsch Gerätebau GmbH, Selb)
Specifications
Samples: liquids, bulk materials and powders
Temperature: RT – 1400 °C
Heating rate: max. 50 K/min (depending on the temperature program)
Sample load: max. 35000 mg (incl. crucible)
Balance resolution: 0.1 µg
Atmospheres: inert, oxidizing, reduzing
Sample crucible: Al2O3 (outer-Ø 6.8 mm, 85 μL); automatic sample changer (20 crucible positions)
Optional: Oxygen analyzer Zirox SGM5T; QMS 403 Aëolos Quadro, amu 300 (SCAN-/MID-mode)
Further details and request for use of equipment
Contact: Dr.-Ing. Jan Gläsel (leitung@tc1.tu-darmstadt.de)
The terms of use (Nutzerordung) can be downloaded here.
The special focus lies on the development of carbon-based electrodes and catalyst. In order to ensure a high reproducibility and highly pure materials, polymers and carbides are employed as the carbon precursors. Particular attention is paid to the precise control of the pore structure from the sub-nanometer towards the mm scale, including spatially varying properties. In this sense, core/shell carbon particles as well as hierarchically structured open cellular materials are prepared. For the later one an unprecedented high degree of freedom is available through a newly developed stereolithographic 3D printing route. For catalytic applications, surface functional groups are introduced or noble and base metals are deposited in controlled amount, size and shape on these carbons by various methods. The in-depth material characterization is the cornerstone in the etzoldlab to connect the resulting material properties with the synthesis procedure and conditions. Among the variety of characterization techniques applied, a special focus is on high-resolution physi- and chemisorption and thermogravimetric analysis coupled with mass spectrometry.
- T. Ariyanto, G.-R. Zhang, F. Riyahi, J. Gläsel and B.J.M. Etzold „Controlled synthesis of core-shell carbide-derived carbons through in situ generated chlorine“. Carbon 115, (2017) 442.
- M. Munoz, S. Ponce, G.-R. Zhang and B.J.M. Etzold„Size-controlled PtNi nanoparticles as highly efficient catalyst for hydrodechlorination reactions“. Appl. Catal. B. 192, (2016) 1.
- T. Ariyanto, B. Dyatkin, G.-Z. Zhang, A. Kern, Y. Gogotsi and B.J.M. Etzold “Synthesis of Carbon Core–Shell Pore Structures and their Performance as Supercapacitors”. Microporous and Mesoporous Mater. 218, (2015) 130.
- J. Gläsel, J. Diao, Z. Feng, M. Hilgart, T. Wolker, D.S. Su and B.J.M. Etzold “Mesoporous and Graphitic Carbide-Derived Carbons as Selective and Stable Catalysts for the Dehydrogenation Reaction”. Chem. Mater. 27, (2015) 5719.
- B.J.M. Etzold, I. Neitzel, M. Kett, F. Strobl, V.N. Mochalin and Y. Gogotsi, “Layer-by-Layer Oxidation for Decreasing the Size of Detonation Nanodiamond”. Chem. Mater. 26, (2014) 3479.
- V.N. Mochalin, I. Neitzel, B.J.M. Etzold, A. Peterson, G. Palmese, and Y. Gogotsi, “Covalent Incorporation of Aminated Nanodiamond into an Epoxy Polymer Network”. ACS Nano 5, 7502 (2011) 7494.
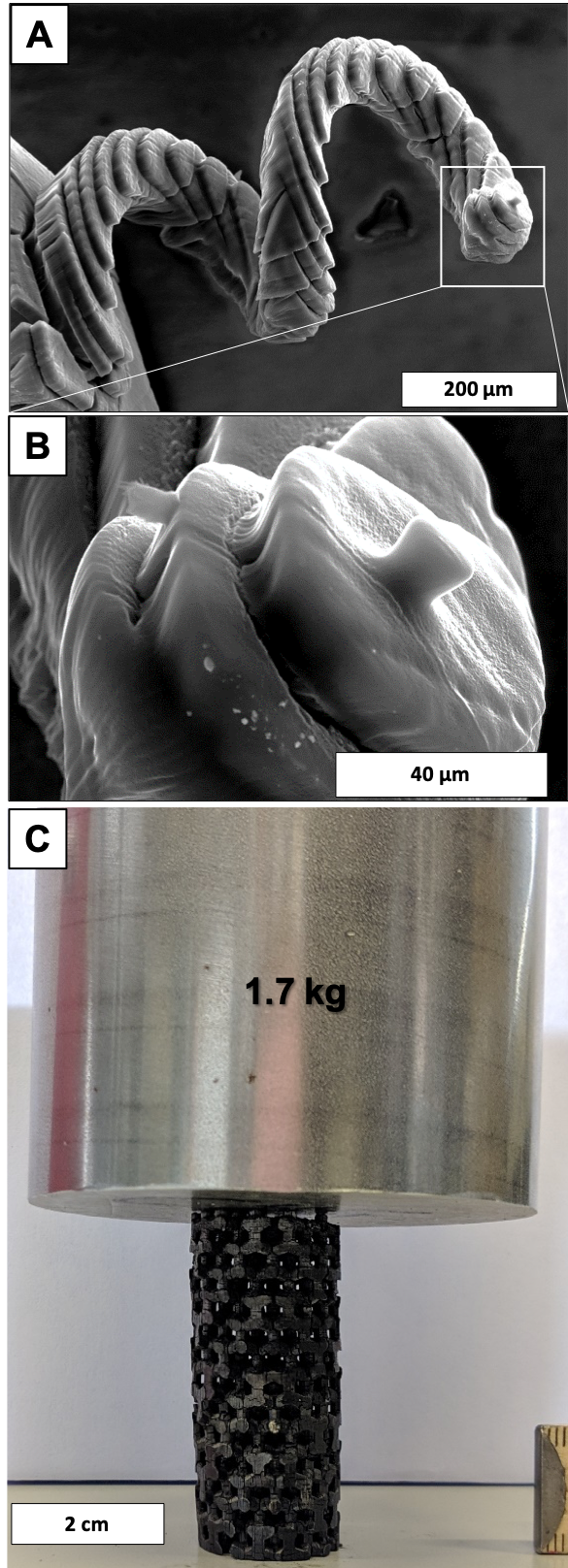
A,B) SEM images of a 3D porous carbon in a spiral macrostructure; C) 3D printed porous carbon open-cell structure supporting the weight of a 1.7 kg steel cylinder.
